Did Your Supplier Have A Poor FDA Inspection?
You’ll always know with Registrar Corp’s FDA Compliance Monitor.
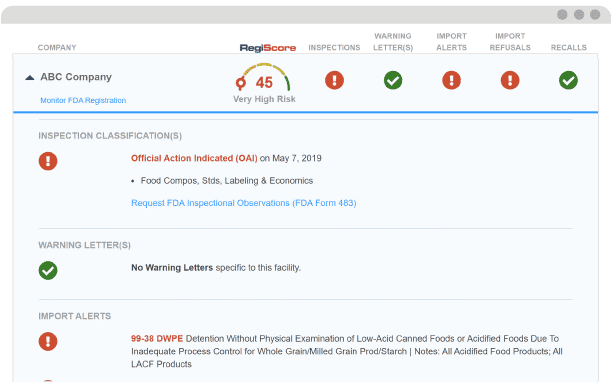
If FDA finds serious regulatory violations at your supplier’s facility, the Agency may hold you liable for importing violative products. Protect your company by tracking your suppliers’ compliance 24/7, including their FDA Inspection Classifications.
Need more information?
Schedule a Demo or Call +1-757-224-0177 option 4.Know Sooner. Act Faster. Reduce Risk.
FDA may inspect your supplier at any time. If FDA finds regulatory violations at your supplier’s facility and you do not properly evaluate them based off this data, the Agency may take enforcement action against you. 24/7 automated supplier checks notify you if your supplier has an FDA inspection.
- Save hours of time on supplier verification activities required by the Foreign Supplier Verification Program (FSVP) rule.
- Take corrective action prior to a shipment to avoid detention costs and enforcement action.
Supply-Chain Management
Track and approve your suppliers in one centralized interface.
Automated Risk Review
Suppliers are scored on a risk scale of 0 to 100 based on their compliance and shipment history
Email Alerts
Receive immediate alerts when a supplier’s compliance changes.
Secure Document Management
Request, review, and store your supplier documentation in a highly secure environment.
Monitor Your Suppliers for FDA Inspections and More
Sign up for an FDA Compliance Monitor account.


